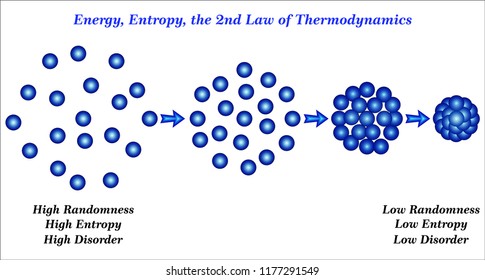
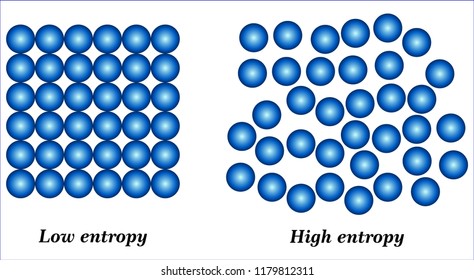
Entropy explained why heat flowed from warm objects to cold ones. Imagine if you dropped your cards and they went flying at the ceiling - that just doesn't make any sense.Ī long time ago scientists (or one in particular, Rudolf Clausis) began to recognize this natural trend towards lower energy and sought to quantify it, sparking the idea of entropy. In fact, the cards fall to the floor in the first place because they would naturally rather go with gravity than oppose it. You've got to repeat this process over and over until all 52 of the cards have been compared and arranged, and that demands a lot of energy. Think about the energy that it takes to arrange cards by value and suit: you've got to look at a card, compare it to others, classify it and then arrange it. The reason a deck of cards doesn't reorganize itself when you drop it is because it's naturally easier for it to remain unordered. So what gives? Why does your desk always get dirty, you ask?Įntropy is a tendency for systems to move towards disorder and a quantification of that disorder. You can't throw a broken egg at the wall and cause it to come back together into its original form, just like your office desk is bound to get messier and messier if you never clean it up. Similarly, when you drop a deck of cards onto the floor they don't arrange themselves by suit or by number (though that would be a nifty trick). Leaves don't fall into neat piles or stack nicely into towers, they just fall. Entropy is even said to be responsible for human life.Įvery fall when the leaves change colour and spill from the trees, they do so randomly. In college, scientists and engineers deal with a similar epiphany when they learn that entropy is the reason that reactions happen, the reason ice melts, the reason that people have to rake up leaves every autumn. How do you describe it? What does it even mean? Who in their right mind would want to quantify a phantom concept that's impossible to see or touch?Īs kids we're usually stunned when we learn that Santa Claus doesn't really exist or that you can't actually bounce around on fluffy white clouds. Journal reference: Physical Review X, DOI: 10.1103/PhysRevX.11.Entropy is a term that draws both fear and reverence from the greatest physicists and mathematicians.
This finding could also help us understand more generally how the quantum world and the classical world are similar and different in terms of thermodynamics and the passage of time. Understanding this relationship could be helpful for designing clocks in the future, particularly those used in quantum computers and other devices where both accuracy and temperature are crucial, says Ares. “It’s still unclear how this principle plays out in real devices such as atomic clocks, which push the ultimate quantum limits of accuracy,” says Mark Mitchison at Trinity College Dublin in Ireland. We can’t be sure yet that these results are actually universal, though, because there are many types of clocks for which the relationship between accuracy and entropy haven’t been tested.

“I think it’s hinting at this universality of how the laws of thermodynamics apply to clocks,” says Ares.
When the researchers compared their results with theoretical models developed for clocks that rely on quantum effects, they were surprised to find that the relationship between accuracy and entropy seemed to be the same for both.
